LC-MS/MS Rapid Toxicology Screening System Ver.3
For LabSolutions™ LCMS
Library info for 231 compounds common in toxicology

This system improves the efficiency of simultaneous LC/MS/MS analysis methods by allowing the user to start analysis without time-consuming steps such as consideration of separation conditions and MS parameter optimization.
Rapid simultaneous analysis methods with analysis times under 15 mins are included for 231 compounds including drugs of abuse, psychoneurotic drugs and sedatives. The system also provides optimised conditions for synchronised survey scans used in screening measurements (conditions adopted for the product ion spectrum as a threshold value for the intensity in MRM).
Total solution including semi-quantitation
For each compound in the method, calibration curve information based on internal standards is available, so it is possible to obtain simple quantitative values after analysis. Using a pretreatment method based on the QuEChERS method*, it is possible to comprehensively recover compounds from biological samples. Qualitative analysis using the spectral library can be carried out during the data processing stage. In other words, the system provides a total solution encompassing pretreatment, analysis and data processing.
Pretreatment using the Micro Volume QuEChERS Kit

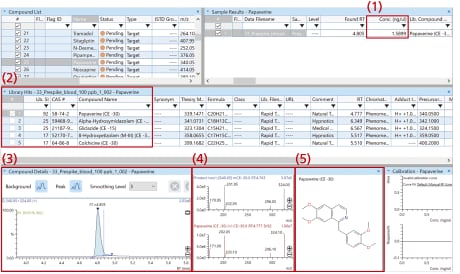
Example of data analysis using LabSolutions Insight Library Screening:
Using LabSolutions Insight Library Screening, you can check all of the following:
- (1) Quantitative results
- (2) Library search results
- (3) MS chromatogram
- (4) MS/MS spectra
- (5) Structural formula
List of registered compounds:
231 compounds including illegal drugs, sedatives, psychotropic drugs and medical drugs associated with many real-life cases.
| IS Diazepam-D5(Positive) IS_Phenobarbital-D5 (negative) 7-Aminoclonazepam 7-Aminoflunitrazepam 7-Aminonimetazepam 7-Aminonitrazepam 8-Hydroxyetizolam (M-III) Acetaminophen Aconitine Alfa-methyltryptamine Allylisopropylacetylurea alpha-Hydroxyalprazolam alpha-Hydroxymidazolam alpha-Hydroxytriazolam Alprazolam Amitriptyline Amlodipine Amobarbital Amoxapine Amphetamine Ampicillin Aripiprazole Atenolol Atomoxetine Atorvastatin Atropine Azelnidipine Azilsartan Barbital Benzoyl ecgonine Biperiden Blonanserin Brexpiprazole Bromazepam Bromocriptine Bromovalerylurea Bromperidol Brotizolam Bupivacaine Caffeine Candesartan Carbamazepine Carbazochrome Carpipramine Carvedilol Chlordiazepoxide Chlorpheniramine Chlorpromazine |
Chlorpromazine-M (bis-nor-) Cibenzoline Clobazam Clocapramine Clomipramine Clonazepam Clotiazepam Cloxazolam Clozapine Cocaine Codeine Colchicine DDVP Delorazepam DEP Desipramine Desmethylclotiazepam Desmethyldiazepam Dextromethorphan Diazepam Dibucaine Diclofenac Dihydrocodeine Diltiazem Diphenhydramine Diprophyline Diquat Domperidone Donepezil Dosulepin Droperidol Duloxetine Ecgonine methyl ester Ephedrine Escitalopram Estazolam Ethenzamide Ethyl loflazepate Etizolam Famotidine Fludiazepam Flufenamic acid Flunitrazepam Fluphenazine Flurazepam Fluvoxamine Furosemide |
Gabapentin Glibenclamide Gliclazide Glimepiride Haloperidol Haloxazolam Hydroxymethylbrotizolam Hydroxyzine Ibuprofen Imidapril Imipramine Irbesartan Isopropylantipyrine Ketamine Ketoprofen Lamotrigine Levetiracetam Levomepromazine Lidocaine Lorazepam Lormetazepam Losartan Loxoprofen Malathion Maprotiline MDA MDMA Medazepam Mefenamic acid Memantine Mepivacaine Mequitazine Metformin Methamphetamine Methomyl Methylephedrine Methylphenidate Mexazolam Mexiletine Mianserin Midazolam Milnacipran Mirtazapine Morphine Mosapramine Naftopidil N-Desmethyl clobazam |
N-Desmethyl zopiclone N-Desmethylmirtazapine Nemonapride Nicardipine Nicotine Nicotine-M Nifedipine Nimetazepam Nitrazepam Norephedrine Nortriptyline Noscapine Olanzapine Olmesartan Oxazepam Oxypertine Paliperidone Papaverine Paroxetine Pemoline Pentazocine Pentobarbital Perospirone Perphenazine Phenobarbital Phenytoin Pimozide Pioglitazone Pipamperone Piroxicam Pitavastatin Pranlukast Primidone Procaine Prochlorperazine Promethazine Propericiazine Propofol Propranolol Quazepam Quetiapine Risperidone Ropivacaine Rosuvastatin Salicylamide Salicylic acid |
Secobarbital Sertraline Setiptiline Sildenafil Silodosin Sitagliptin Solifenacin Spiperone Spironolactone Sulfamethoxazole Sulpiride Sultopride Suvorexant Tadalafil Tandospirone Telmisartan Temazepam Tetracaine THC THC-COOH Thiamylal Timiperone Tofisopam Topiramate Tramadol Trandolapril Trazodone Triazolam Trihexyphenidyl Trimethoprim Trimipramine Urapidil Valproic Acid Valsartan Vardenafil Venlafaxine Verapamil Warfarin Zalepron Zolpidem Zolpidem _M-1 Zonisamide Zopiclone Zopiclone-N-oxide Zotepine |
Remarks and Precautions
- LabSolutions LCMS Ver. 5.109 or later and LabSolutions Insight Ver. 3.8SP1 or later are required.
- Depending on the condition and specifics of the equipment used, differences in quantitative values may occur. For accurate quantitative values, please use a calibration curve method with appropriate standard samples. In addition, scores obtained from searching in the library show the similarity of spectra and form one basis for the identification of compounds, but for more accurate compound identification it is more effective to combine multifaceted information such as retention times and sample information.
LabSolutions is a trademark of Shimadzu Corporation or its affiliated companies in Japan and/or other countries.
For Research Use Only. Not for use in diagnostic procedures.



